News on health and wellness in Oregon
Provided by AGPSilexion Therapeutics Reports Positive Preliminary Immunotherapy Findings for SIL204 in KRAS-Driven Pancreatic Cancer
Preliminary results from a preclinical study demonstrated statistically significant increase in MHC-I expression following SIL204 treatment in human pancreatic and non-small cell lung cancer cells harboring a KRAS mutation, supporting potential future evaluation alongside anti-PD-1 therapies including pembrolizumab (Keytruda®)
Grand Cayman, Cayman Islands, May 14, 2026 (GLOBE NEWSWIRE) -- Silexion Therapeutics Corp. (NASDAQ: SLXN) ("Silexion" or the "Company"), a clinical-stage biotechnology company pioneering RNA interference (RNAi) therapies for KRAS-driven cancers, today announced positive preclinical findings from an ongoing translational immuno-oncology study evaluating its lead candidate, SIL204, in human KRAS-mutated pancreatic cancer cells. The study demonstrated a statistically significant increase in surface expression of major histocompatibility complex class I (MHC-I), also known as HLA-ABC, following treatment with SIL204 in human pancreatic cancer cells harboring the KRAS G12R mutation, as measured by flow cytometry.
“These findings are particularly encouraging because they suggest SIL204 may influence biological pathways involved in the tumors evading the immune cells which are supposed to protect against the tumors, in addition to its previously demonstrated direct anti-tumor activity,” said Ilan Hadar, Chairman and Chief Executive Officer of Silexion Therapeutics. “Checkpoint inhibitors have historically shown limited efficacy in pancreatic cancer in part because T cells often fail to adequately recognize these tumors. We believe the observed increase in MHC-I expression further supports an additional positive role of SIL204 in the area of immunotherapy which could facilitate positive outcomes in the treatment of pancreatic cancer.”
MHC-I is essential for enabling cytotoxic T cells to recognize and attack tumor cells. Loss or suppression of MHC-I expression is widely recognized as a key mechanism by which tumors evade immune detection and resist immune-mediated destruction. Research has shown that oncogenic KRAS signaling contributes to immune evasion through suppression of antigen presentation and impairment of T-cell recognition pathways in pancreatic cancer and other KRAS-driven tumors.
Pancreatic cancer remains among the most immunologically resistant solid tumors and has historically demonstrated limited responsiveness to immune checkpoint inhibitor therapies such as anti-PD-1 agents, including pembrolizumab (Keytruda®), outside of select biomarker-defined patient populations. By increasing MHC-I expression, SIL204 may help restore immune visibility of KRAS-mutated tumor cells, potentially supporting future therapeutic strategies designed to enhance responsiveness to PD-1/PD-L1 blockade.
Published research has increasingly highlighted the relationship between KRAS signaling, antigen presentation, and immune checkpoint resistance, with multiple recent studies suggesting that reversing KRAS-associated immune suppression may improve immune-mediated anti-tumor activity.1
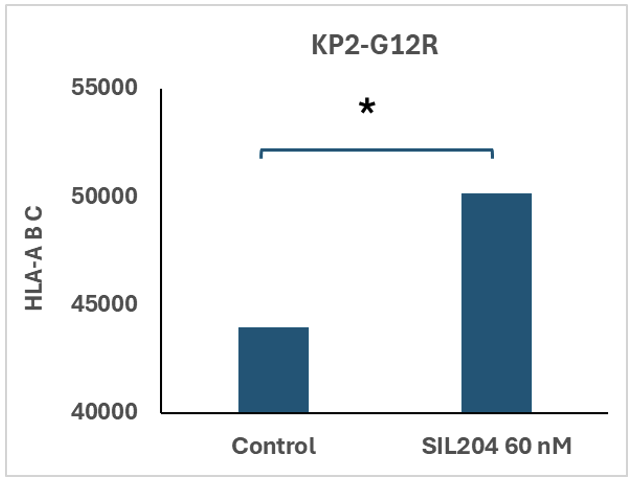
Image Caption: SIL204 increases surface MHC-I (HLA-ABC) expression in human KRAS G12R-mutated pancreatic cancer cells (KP2-G12R) at 60 nM, as measured by flow cytometry. *P<0.05 vs. control.
About Silexion Therapeutics
Silexion Therapeutics is a pioneering clinical-stage, oncology-focused biotechnology company dedicated to the development of innovative treatments for unsatisfactorily treated solid tumor cancers that have the mutated KRAS oncogene, generally considered to be the most common oncogenic gene driver in human cancers. The Company conducted a Phase 2a clinical trial in its first-generation product, which showed a positive trend in comparison to the control of chemotherapy alone, and is currently advancing its lead, second-generation, product candidate, SIL204, a small interfering RNA (siRNA), towards clinical trials in Israel and the European Union. Silexion is committed to pushing the boundaries of therapeutic advancements in the field of oncology and further developing its lead product candidate for locally advanced pancreatic cancer. For more information, please visit: https://silexion.com
Notice Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the federal securities laws. All statements other than statements of historical fact contained in this communication, including statements regarding the therapeutic potential, immune-modulating activity, mechanism of action, translational significance, future development, and planned studies relating to SIL204, including its potential role in modulating antigen presentation, restoring immune recognition, enhancing responsiveness to checkpoint inhibitor therapies, and future immuno-oncology or combination applications, are forward-looking statements. These forward-looking statements are generally identified by terminology such as "may", "should", "could", "might", "plan", "expect", "intend", "will", "estimate", "anticipate", "believe", "predict", or "potential", or the negatives of these terms or variations of them or similar terminology. Forward-looking statements involve a number of risks, uncertainties, and assumptions, and actual results or events may differ materially from those projected or implied in those statements. Important factors that could cause such differences include, but are not limited to: (i) Silexion's ability to successfully complete preclinical studies and initiate and conduct clinical trials, including the Phase 2/3 trial of SIL204 in locally advanced pancreatic cancer; (ii) Silexion's strategy, future operations, financial position, projected costs, prospects, and plans; (iii) the impact of the regulatory environment and compliance complexities, including the outcome of the CTA’s review of the Company’s application to commence clinical trials in Germany and other jurisdictions, as well as site-level approvals, conditions and clearances (including outstanding regulatory forms and any initial participant caps) required prior to study commencement at each clinical site; (iv) expectations regarding future partnerships or other relationships with third parties; (v) Silexion's future capital requirements and sources and uses of cash, including its ability to obtain additional capital; (vi) Silexion's ability to maintain its Nasdaq listing; and (vii) other risks and uncertainties set forth in the documents filed by the Company with the SEC, including the Company's Annual Report on Form 10-K for the year ended December 31, 2025. Silexion cautions you against placing undue reliance on forward-looking statements, which reflect current beliefs and are based on information currently available as of the date a forward-looking statement is made. Forward-looking statements set forth herein speak only as of the date they are made. Silexion undertakes no obligation to revise forward-looking statements to reflect future events, changes in circumstances, or changes in beliefs, except as otherwise required by law.
Company Contact
Silexion Therapeutics Corp
Ms. Mirit Horenshtein Hadar, CFO
mirit@silexion.com
Investor Relations Contact
Arx Investor Relation
North American Equities Desk
silexion@arxhq.com
1 See Bear AS et al., Cancer Cell (2020); Canon J et al., Nature (2019)

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.

